Sildenafil and other phosophodiesterase-5 inhibitors(PDE-5 inhibitors) have taken the drug market by storm for erectile dysfunction. Viagra-just to name one of the drug names- has certainly enhanced the treatment possibilities for a male problem that was difficult to treat in the past. The market has had its heyday with splashy TV commercials, creating hype like seldom before. The truth remains, that any drug has also possible side effects that are not mentioned in the upbeat commercials, and patients with preexisting heart disease have experienced heart attacks. The medications for the treatment of erectile dysfunction are not recreational fun, but serious prescription drugs.
In the meantime these drugs are facing competition from a new class of erectile dysfunction drugs, the melanocortin agonists.
They claim to have remarkable properties due to the fact that they will not only help men but also women with a range of sexual disorders, including lack of desire. Contrary to the previous drugs they are not working through the vascular system but through the central nervous system. Through receptors in the brain area called hypothalamus, they stimulate areas of the brain associated with sexual arousal. Preliminary experiments have shown that they are increasing libido, but also help a man to get better erections.
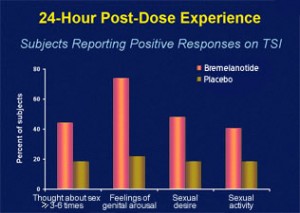
The melanocortin agonist closest to the market is known as bremelanotide. Previously known as PT-141, the nasal spray has been tested in about 300 men up to phase II with promising results. A study on women has been too small in numbers to give detailed results, but significantly more women reported increased libido after bremelanotide treatment as opposed to placebo. The drug’s half-life is only two hours, but women have reported effects lasting 10 to 12 hours. They also reported that the quality of their sexual encounter had improved. The older group (women over 34) responded slightly better than their younger counterparts.
Bremelanotide still has some way to go till it will be on the market, but it is getting some positive press in magazines as the “first equal-opportunity aphrodisiac”. Palatin Technologies, the manufacturer of the drug is more cautious and does not want bremelanotide to be perceived as a leisure drug, even more so as it has not seen its approval at this point.
National Review Of Medicine, July 30, 2006, page 11
Comment on Nov. 13, 2012: The FDA did not approve this drug due to reports of elevated blood pressure in a minority of test subjects. However, the drug has been synthetized by other pharmaceutical companies around the globe and is actively traded in Argentina, Armenia, Aruba, Australia, Austria, Azerbaijan, the Bahamas, the United Arab Emirates, United Kingdom and Uruguay.
Last edited December 6, 2012